Like most people, you probably use your TV’s remote control, have a wall clock, or turn on a flashlight, without giving much thought as to how they work…until now. Exploring the science behind what makes your TV’s remote allow you to fire through channels like the beat of your heart, may seem daunting. However, a peak inside a battery can help you understand and appreciate the magic that happens when battery power is connected to your favorite device.
Anatomy of a Battery
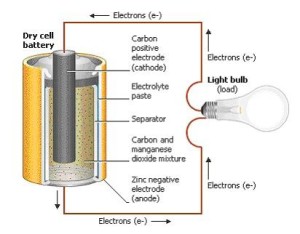
Two important terminals exist inside every battery: one positive (+) and one negative (-). You can see the positive and negative signs on the ends in common batteries such as AA type.
These other internal parts of a battery work together to help produce electricity.
Cathode – The cathode, an electrode, is connected with the positive terminal. (An electrode is a an electrical conductor).
Anode – The anode, also an electrode, is connected with the negative terminal.
Separator – The separator keeps the cathode and the anode from touching.
Electrolyte – The electrolyte is a liquid that permits an electrical charge to flow between the cathode and the anode.
Collector – The collector carries the charge outside the battery and through the device needing power. For instance, a portable radio or a light bulb.
Power-up!
When you correctly insert a battery into a flashlight or other device, chemical reactions occur at both the cathode and the anode to make electricity. In short, the reactions at the anode create electrons, while the reactions at the cathode receive them. Let’s examine this idea a bit more closely.
 During the anode reaction, two or more electrically charged atoms or molecules (called ions) from the electrolyte join with the anode to make a compound. As this happens, one or more electrons are released. This activity is known as an oxidation reaction.
During the anode reaction, two or more electrically charged atoms or molecules (called ions) from the electrolyte join with the anode to make a compound. As this happens, one or more electrons are released. This activity is known as an oxidation reaction.
During the cathode reaction, ions combine with both material from the cathode and the free electrons to make compounds. This activity is known as a reduction reaction. The combined reactions generate the flow of electricity that powers your gadget, tool or device.
A battery provides power until either the cathode or the anode, or both, has no more material that makes the reactions occur. Next time your battery runs out, you’ll know why.
You’ll also like:
Are you Crazy to want a Battery Backup system for your Solar Panels?
No Pests, No Dirt, No Worries…Bring the Garden Indoors!
Fuel Neighbor Envy with Stunning LED Light Strips

Leave a Reply